Letter: Reactions in Molecular Clusters: Proton Transfer to Small Amino Acids - Anja Meffert, Jürgen Grotemeyer, 1995

Nitrogen Dioxide Reaction with Nitroxide Radical Derived from Hydroxamic Acids: The Intermediacy of Acyl Nitroso and Nitroxyl (HNO) | The Journal of Physical Chemistry A
Thermodynamics of multicomponent, miscible, ionic systems: theory and equations | The Journal of Physical Chemistry

Full-dimensional quantum stereodynamics of the non-adiabatic quenching of OH(A2Σ+) by H2 | Nature Chemistry
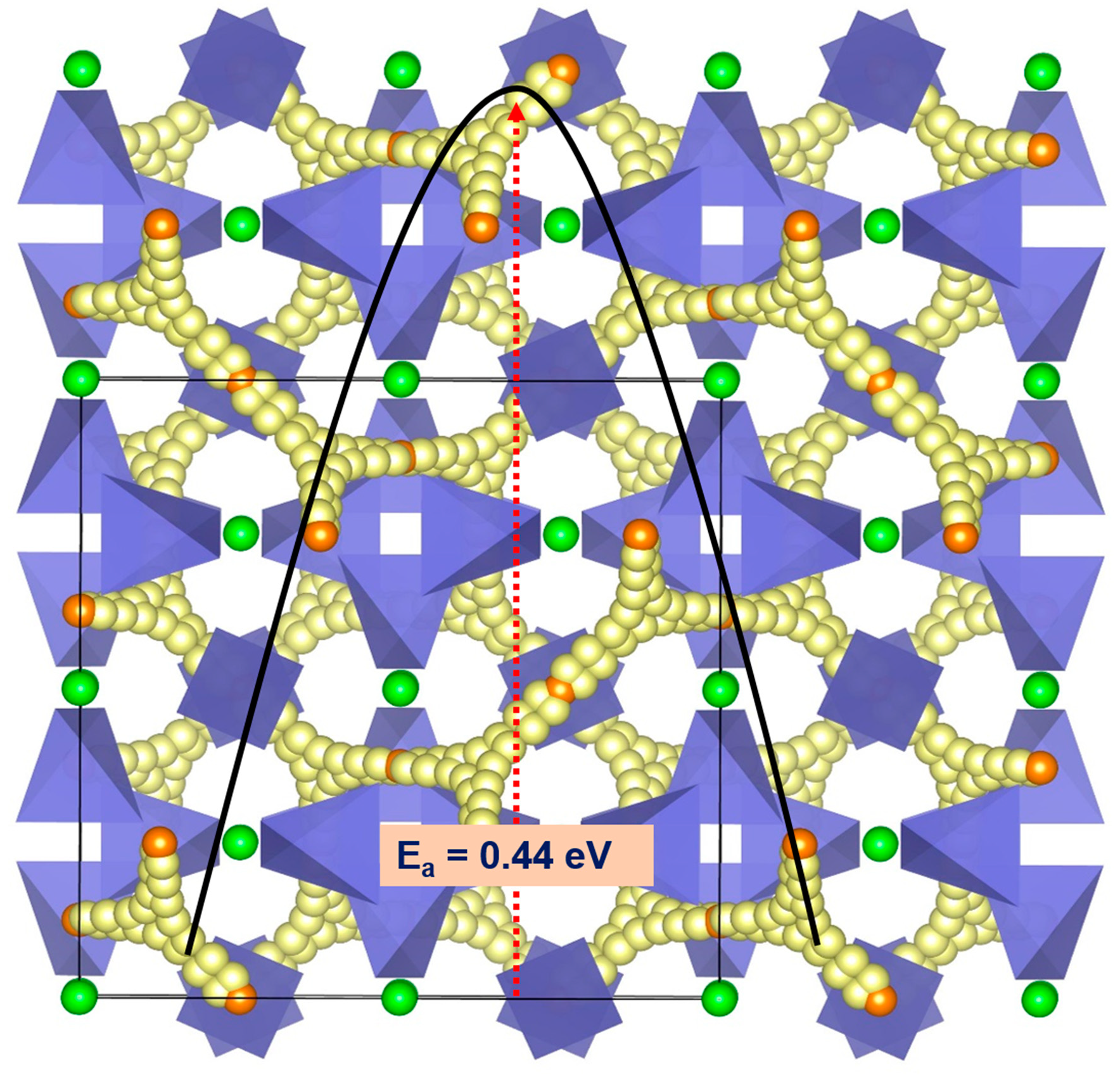
Physchem | Free Full-Text | Computational Study of Crystallography, Defects, Ion Migration and Dopants in Almandine Garnet
Detailed photocurrent spectroscopy of the semiconducting group VIB transition metal dichalcogenides | The Journal of Physical Chemistry

Q‐Chem 2.0: a high‐performance ab initio electronic structure program package - Kong - 2000 - Journal of Computational Chemistry - Wiley Online Library

Full article: Time-dependent dynamics of nuclear spin symmetry and parity violation in dichlorodisulfane (ClSSCl) during and after coherent radiative excitation

PDF) Structure and spectroscopy of (HCN)n clusters: Cooperative and electronic delocalization effects in CH . . . N hydrogen bonding
Bipolar cadmium selenide/cobalt(II) sulfide semiconductor photoelectrode arrays for unassisted photolytic water splitting | The Journal of Physical Chemistry
Single-Molecule Fluorescence Analysis in Solution - Richard A. Keller, W. Patrick Ambrose, Peter M. Goodwin, James H. Jett, John C. Martin, Ming Wu, 1996
Counter-current diffusion of silver nitrate and hydrochloric acid in cellulose and Nafion films | The Journal of Physical Chemistry

Large Quadratic Hyperpolarizabilities with Donor–Acceptor Polyenes Exhibiting Optimum Bond Length Alternation: Correlation Between Structure and Hyperpolarizability - Blanchard‐Desce - 1997 - Chemistry – A European Journal - Wiley Online Library

Theory of hydrogen bonding in supercritical fluids - Gupta - 1992 - AIChE Journal - Wiley Online Library

Theory of Optical Spectra of Photosystem II Reaction Centers: Location of the Triplet State and the Identity of the Primary Electron Donor: Biophysical Journal
Microphase Formation in Aqueous Solutions of an Ethylene Oxide/Propylene Oxide Blockcopolymer - H. Hespe, J. Crone, E.H. Mueller, E.E. Schaefer, 1982





