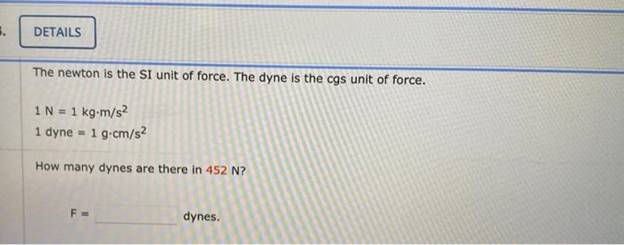
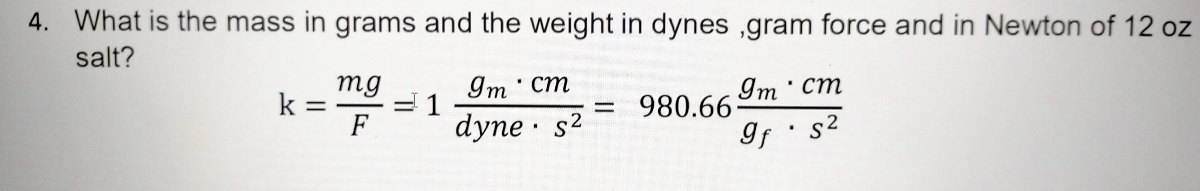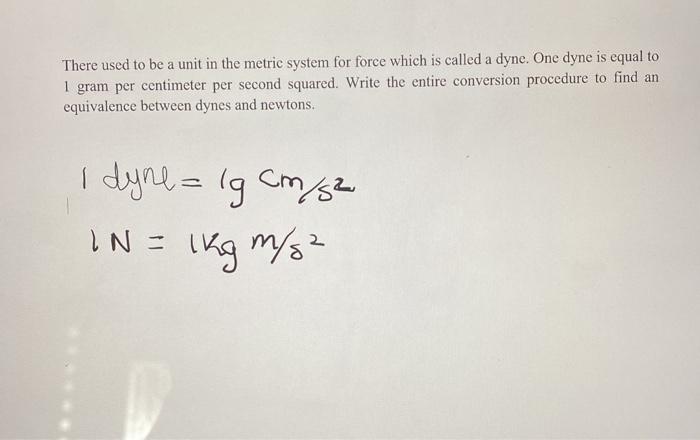Activity 1: solve for size and speed of a drop.. Useful units for calculations Remember: dyne = 1 g cm/s 2 (force in cm-g-s units) = Newtons. Using. - ppt download

Dyne Espresso Flavoured Freeze Dried Instant Coffee 60 Gram Jar (Cold Coffee and Hot Coffee) : Amazon.in: Grocery & Gourmet Foods
The value of G in C.G.S system is 6.67 × 10^-8 dyne cm^2 g^-2. Calculate its value in S.l. system. - Sarthaks eConnect | Largest Online Education Community
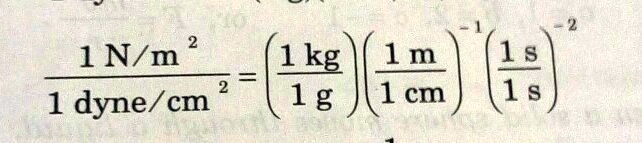
SOLVED: Solve it, please. Conversion factor between newton and dyne: 1 N/m = 1 dyne/cm. 1 kg = 1 N. (4 g cm)/(1 )

What will be the pressure in dyne cm^-2 , due to a water column of height 12.5cm ? ( g = 980cm s^-2 ) (density of water 10^3kg m^-3 ).

SOLVED:If at a pressure of 10^6 dyne / cm^2, one gram mole of nitrogen occupies 2 ×10^4 c.c. volume, then the average energy of a nitrogen molecules in erg is : (Given:

The value fo universal gravitationla constant `G` in `CGS` system is `6.67xx10^(-8)` dyne `cm^(2) - YouTube

One - gram mole of nitrogen occupies 2 × 10^4 cc at a pressure of 10^6 dynes/cm^2 . The average energy of a nitrogen molecule (in erg) will be: (Avogadro's number = 6 × 10^25 )

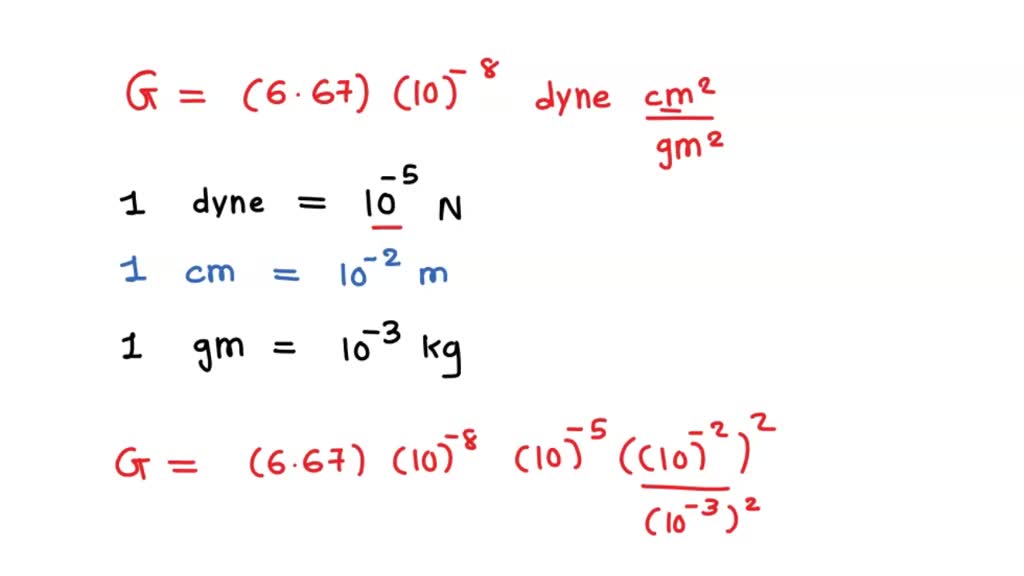
![Bengali] What is the value of bar in dyne//cm^2 unit. Bengali] What is the value of bar in dyne//cm^2 unit.](https://d10lpgp6xz60nq.cloudfront.net/ss/web-overlay-thumb/9094290.webp)