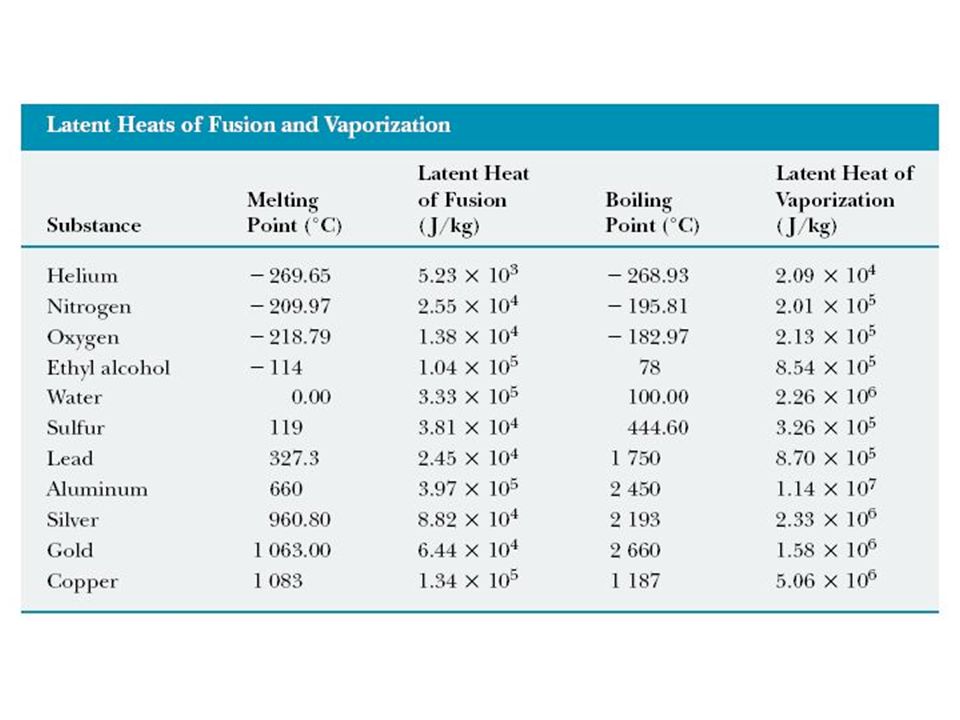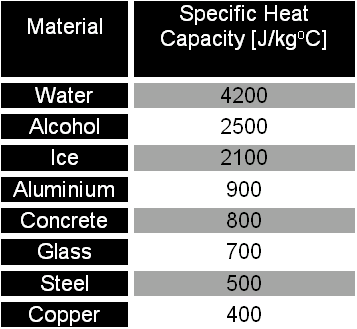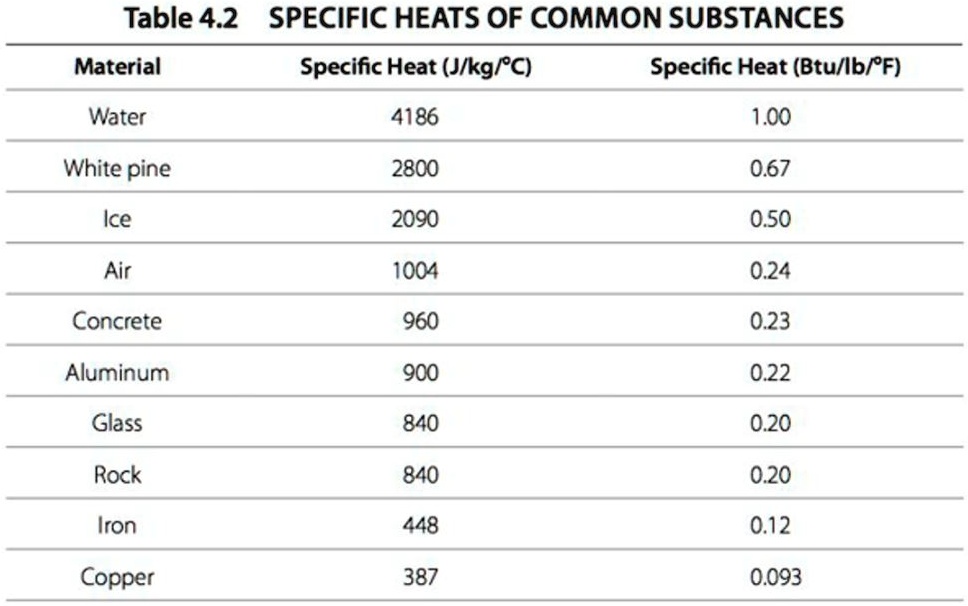
SOLVED: Table 4.2 SPECIFIC HEATS OF COMMON SUBSTANCES Material Specific Heat (JlkgrC) Specific Heat (BtullbrF) Water 4186 1.00 White pine 2800 0.67 Ice 2090 0.50 Air 1004 0.24 Concrete 960 0.23 Aluminum 900 0.22 Glass 840 0.20 Rock 840 0.20 Iron 448 ...

Specific Heat Capacity Measurement for Porous Composite Material with Glass Fiber/Fumed Silica Nanoparticles | Semantic Scholar

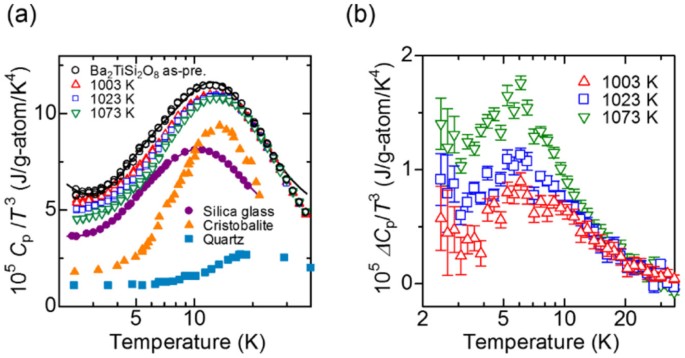
Heat capacity (C p ) vs. temperature curves in the glass transition... | Download Scientific Diagram
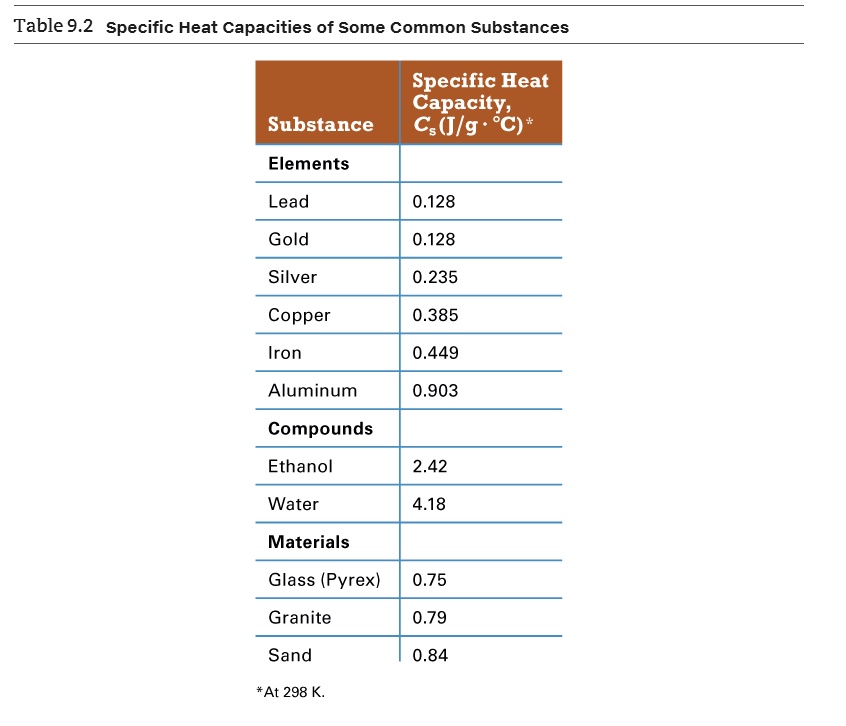
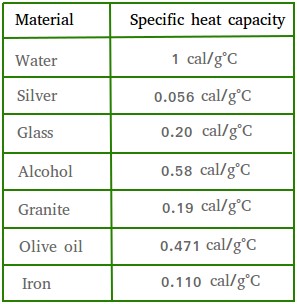
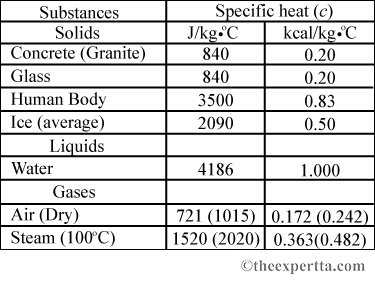
SOLVED: Table 9.2 Specific Heat Capacities of Some Common Substances Specific Heat Capacity; Cs(Jlg. *C) Substance Elements Lead 0.128 Gold 0.128 Silver 0.235 Copper 0.385 Iron 0.449 Aluminum 0.903 Compounds Ethanol 2.42

Specific heat and magnetization studies of spin-glass like transition in nanogranular Cu90Co10 ribbon - ScienceDirect

Specific heat and magnetization studies of spin-glass like transition in nanogranular Cu90Co10 ribbon - ScienceDirect

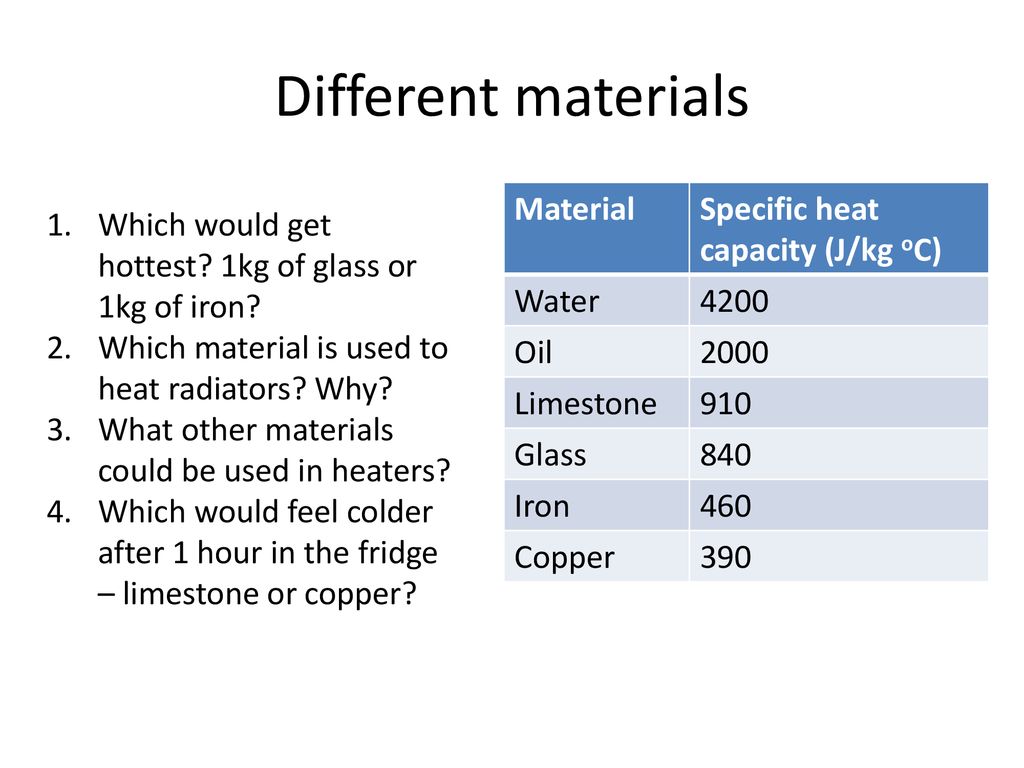
Two bars of identical mass are at 29 degrees C. One is made from glass and the other from another substance. The specific heat capacity of glass is 840 J/(kgC). When identical
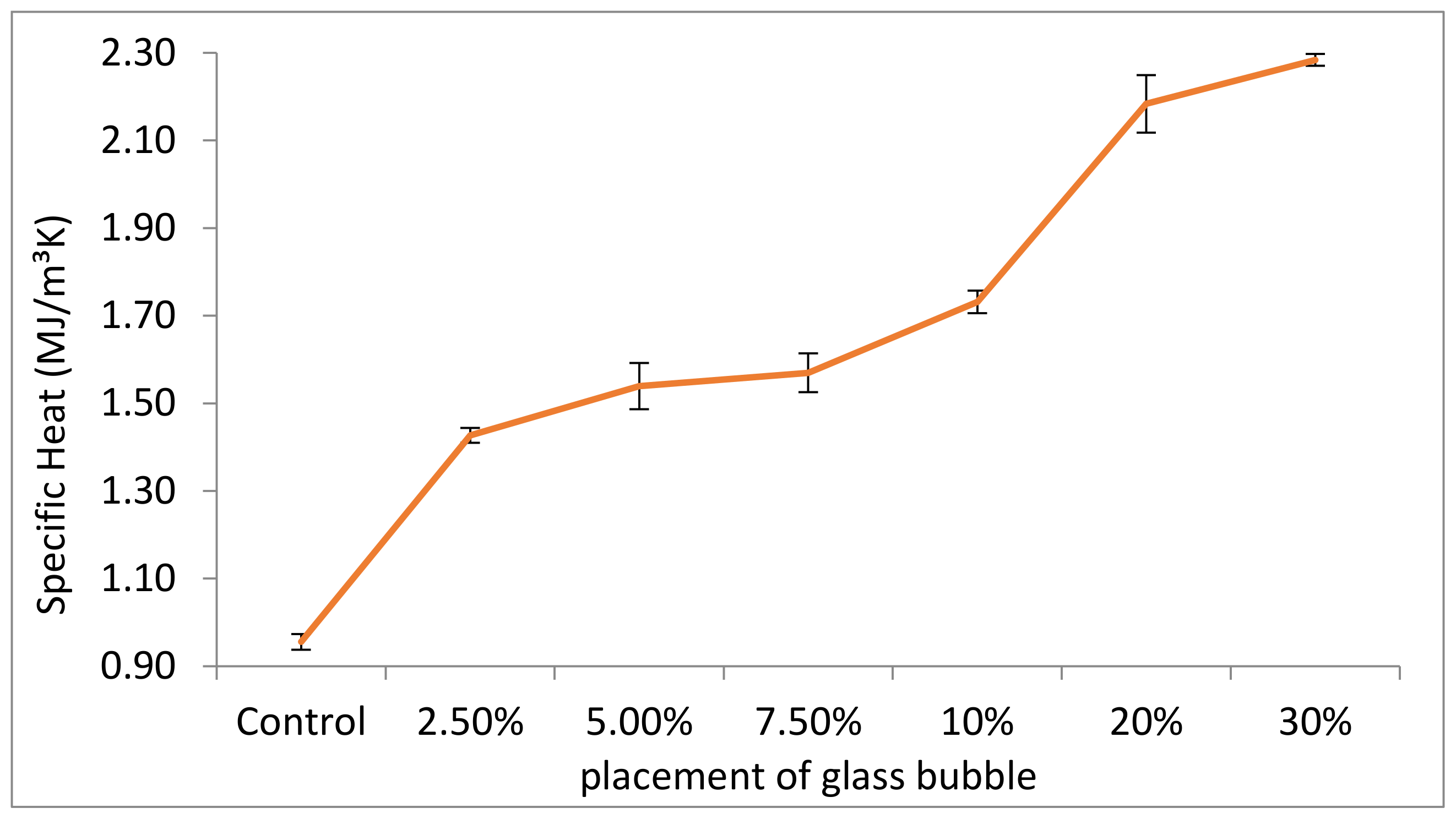
Materials | Free Full-Text | Properties of a New Insulation Material Glass Bubble in Geopolymer Concrete